It is best to be patient at this stage!Īfter pouring the separating gel (ensuring you’ve left enough space for the stacking gel and comb) fill up the empty space with hydrated isopropanol whilst it is polymerizing. If a gel hasn’t properly polymerized this could lead to "wavy" bands. I have found that making fresh 10% APS improved the polymerization of my gels, and it doesn’t take long to prepare. Usually I prepare slightly more stacking gel and separating gel, therefore I can check the tube in which I have prepared the solutions to see if they have polymerized rather than manipulating the gel itself. Before starting to prepare your gel, check the molecular weight of your proteins and decide which gel percentage is most appropriate. If your protein of interest has a low molecular weight and your acrylamide percentage is too low, you risk it running off the gel. The acrylamide percentage you use is proportional to how easy it is for proteins to move through the gel. Make sure your solutions are made up correctly and are at the right pH the stacking gel is usually acidic at pH 6.8 and the separating gel is alkaline at pH 8.8. Sometimes, hours of work will have been put into preparing samples prior to carrying out the western blot but if the gel is not properly prepared all your hard work might go to waste, which would be a massive shame! Moreover, make sure you are always wearing gloves and all other equipment e.g. One way to prevent background is to clean all plates and combs with either 1% SDS or 70% Ethanol followed by distilled water then wipe them dry.

And, because life is unfair, to me the background always seemed to coincide with where my bands should be therefore interfering with any subsequent quantification. Any dirt, dust or leftover gel on your plates or membrane WILL show up as background on your blots. This cannot be emphasized heavily enough. This prompted me to do a lot of troubleshooting along the road to a perfect western, and here I will provide some tips on how to achieve this. It definitely wasn’t as easy as I expected – I was getting different results with every blot and my blots were ALWAYS full of speckles in the background.
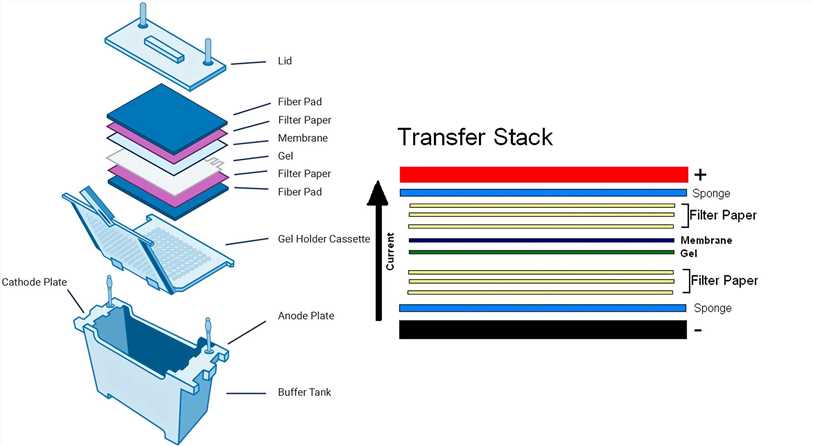
In the first few months of my PhD I was attempting a lot of western blots (something which I had only read about before, I mean, how hard could it be to get a nice single band on a clean blot?). The membrane can then be scanned to visualize the protein of interest. The protein of interest is probed for using a specific antibody and finally detected by a secondary antibody, often conjugated to a fluorophore. The proteins are then transferred to a solid membrane, either nitrocellulose or PVDF, followed by a blocking step to prevent antibodies non-specifically binding to the membrane. Firstly, samples are separated by their molecular weight via gel electrophoresis. This is a technique utilizing antibodies to identify a protein of interest from a mixture in a biological sample. Scientists often wish to study proteins and one way to achieve this is by western blotting. So I don't understand how the protein could either come out the other side, or not go in at all, but the ladder be fine.Proteins are the molecular machinery of a cell, ensuring that every cell in the body can carry out its specific role. Would one of the two observations listed above suggest something that would allow the pre-stained ladder to transfer successfully, but not the protein? The ladder transfered evenly from 250 all the way down to 15 kD.

So there is no protein, but the curious thing is the pre-stained ladder came out fine. pvdf thread in this forum that not soaking in water long enough can prevent protein transfer in PVDF)Ģ) His transfer chamber, when put at the same voltage I normally run (30v) went up to something like 260 mA which is about 4 times higher than the normal mA I see with my mini-transfer chamber. I told him to pre-wet in methanol, which we did, but instead of soaking in water for 5 minutes as per the instructions, he just rinsed and put in the transfer sandwich. When he was putting together the transfer, he dunked the pvdf directly in water and acted surprised when it didnt hydrate. Two suspicious things:ġ) This professor always used nitrocellulose and we use PVDF (hybond-P). In fact, except for the sample preparation in sample buffer, he pretty much did everything. I ran the big gel with professor who does them a lot. However, I wanted to get more resolution so for this particular western I had run a "big" gel. In fact, I had run these exact samples already twice and it worked fine.

I run mini-gels all the time and never have problems. I encountered something strange the other day with my western blot:Īfter transfer, the pre-stained ladder (bio-rad) showed up fine, but there was no protein whatsoever on the blot stained with Ponceau.

